Page 180 - ICSE Chemistry 8
P. 180
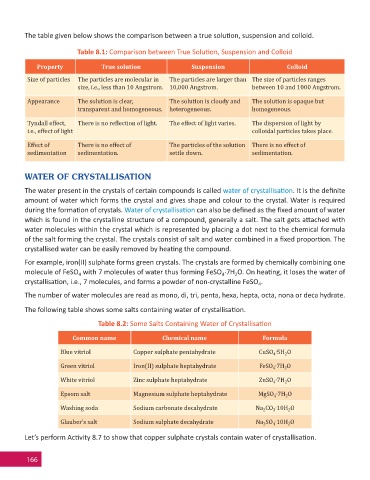
The table given below shows the comparison between a true solu on, suspension and colloid.
Table 8.1: Comparison between True Solu on, Suspension and Colloid
Property True solution Suspension Colloid
Size of particles The particles are molecular in The particles are larger than The size of particles ranges
size, i.e., less than 10 Angstrom. 10,000 Angstrom. between 10 and 1000 Angstrom.
Appearance The solution is clear, The solution is cloudy and The solution is opaque but
transparent and homogeneous. heterogeneous. homogeneous.
Tyndall effect, There is no re lection of light. The effect of light varies. The dispersion of light by
i.e., effect of light colloidal particles takes place.
Effect of There is no effect of The particles of the solution There is no effect of
sedimentation sedimentation. settle down. sedimentation.
WATER OF CRYSTALLISATION
The water present in the crystals of certain compounds is called water of crystallisa on. It is the defi nite
amount of water which forms the crystal and gives shape and colour to the crystal. Water is required
during the forma on of crystals. Water of crystallisa on can also be defi ned as the fi xed amount of water
which is found in the crystalline structure of a compound, generally a salt. The salt gets a ached with
water molecules within the crystal which is represented by placing a dot next to the chemical formula
of the salt forming the crystal. The crystals consist of salt and water combined in a fi xed propor on. The
crystallised water can be easily removed by hea ng the compound.
For example, iron(II) sulphate forms green crystals. The crystals are formed by chemically combining one
.
molecule of FeSO with 7 molecules of water thus forming FeSO 7H O. On hea ng, it loses the water of
4
4
2
crystallisa on, i.e., 7 molecules, and forms a powder of non-crystalline FeSO .
4
The number of water molecules are read as mono, di, tri, penta, hexa, hepta, octa, nona or deca hydrate.
The following table shows some salts containing water of crystallisa on.
Table 8.2: Some Salts Containing Water of Crystallisa on
Common name Chemical name Formula
.
Blue vitriol Copper sulphate pentahydrate CuSO 5H O
2
4
.
Green vitriol Iron(II) sulphate heptahydrate FeSO 7H O
2
4
.
White vitriol Zinc sulphate heptahydrate ZnSO 7H O
2
4
.
Epsom salt Magnesium sulphate heptahydrate MgSO 7H O
4
2
.
Washing soda Sodium carbonate decahydrate Na CO 10H O
3
2
2
.
Glauber’s salt Sodium sulphate decahydrate Na SO 10H O
2
2
4
Let’s perform Ac vity 8.7 to show that copper sulphate crystals contain water of crystallisa on.
166